The Amazing Crystallization ExperimentIn nature it can take eons for crystals to form. How would you like to watch them appear right before your very eyes? |
MICROSCOPE LESSON 6 |
CAUTION: Salicylic acid solution contains rubbing alcohol. Don't swallow it or get it in your eyes. If it gets on your skin or clothing, rinse it off.
|
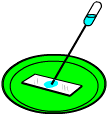
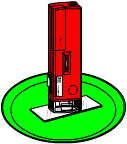
What To Do: Set the slide on the colored plate, which will make a good background for your crystals. With the vial tightly capped, shake the solution for about 20 seconds. (Cap the vial when you're not using it so the alcohol doesn't evaporate.) Using the thin pipette, drip a single tiny drop of solution onto the middle of the slide. It should spread out to about an inch wide, then start to dry from the outside edges. As it dries, place the microscope where you can see some dry and some wet. From this point of view you can see an amazing thing: the formation of crystals!
When the drop is completely dried, you can repeat the experiment by putting another tiny drop right where you put the first drop. |
Once your crystals have formed, you can get a better view of them by using your microscope to look through the slide toward a light source behind it. Keep the microscope light on, too. | |